Last updated: May 09, 2026
The Apple Watch ECG works by completing a Lead I electrical circuit between two electrodes — the watch case back on your left wrist and the Digital Crown touched by a right-hand finger — then sampling the resulting ~1 mV voltage difference at roughly 512 Hz for 30 seconds. An on-device classifier examines R-R interval irregularity and P-wave morphology and returns one of six labels: sinus rhythm, atrial fibrillation, low heart rate, high heart rate, inconclusive, or poor recording. The FDA cleared this pathway under De Novo DEN180044 on September 11, 2018, with K201525 later extending the rhythm window from 50–120 BPM to 50–150 BPM.
The Apple Watch ECG records 30 seconds of single-lead Lead I between two electrodes — the case back on your wrist, the Digital Crown touched by your other hand — and an on-device classifier returns sinus rhythm, AFib, low/high heart rate, inconclusive, or poor recording. The FDA cleared it under De Novo DEN180044 in September 2018 for AFib screening only, in adults 22 and older, with no pacemaker or ICD. A clean strip is screening data, not a diagnosis: export the PDF and bring it to a clinician before acting on it.
In this post
- Lead I on a wrist: the bioelectrical geometry that makes a watch + finger work
- The signal chain from skin to label
- What DEN180044 actually cleared — and what it didn’t
- PPG vs ECG: two sensors, two features, one common confusion
- Why “inconclusive” is the correct answer
- Decision framework: when to trust the watch, when to repeat, when to escalate
- Reading a real strip: what the PDF export shows
- What changed Series 4 → Ultra 2, and what didn’t
- Further reading
- FDA cleared the ECG app via De Novo DEN180044 on September 11, 2018, for users aged 22 and older.
- The recording is single-lead Lead I — left-arm minus right-arm potential — sampled at roughly 512 Hz for 30 seconds.
- Classification runs on-device; possible labels are sinus rhythm, AFib, low or high heart rate, inconclusive, or poor recording.
- The detection window expanded from 50–120 BPM (v1) to 50–150 BPM (v2 clearance, K201525).
- The app is contraindicated for users with pacemakers or ICDs and is not designed to detect heart attack, stroke, blood clots, or any arrhythmia other than AFib.
Lead I on a wrist: the bioelectrical geometry that makes a watch + finger work
An electrocardiogram measures voltage differences across the body produced by depolarization waves traveling through cardiac muscle. Willem Einthoven’s 1903 system used three limb leads, with Lead I defined as the potential difference between the left arm and the right arm, positive at the left arm. Wear an Apple Watch on your left wrist, place a finger from your right hand on the Digital Crown, and the closed circuit is exactly that: positive electrode on the left wrist, negative reference at the right hand. The signal path is electrically identical to a clinician’s Lead I.
That geometry is what defines the device’s scope. A 12-lead ECG uses ten electrodes to triangulate vectors across the frontal and horizontal planes, which is how clinicians localize ischemia, identify hypertrophy, or characterize bundle branch blocks. With only Lead I, you see the projection of the cardiac vector along a single horizontal axis. P-wave morphology, QRS timing, and R-R intervals are all visible — enough to tell whether atrial activity is organized and whether ventricles fire on time. ST-elevation localization, posterior MI patterns, and most ectopic-focus identification are not.

The diagram above shows the closed-loop geometry: skin contact at two points connects across the chest through tissue impedance, and the watch reads the resulting micro-voltage. If a user wears the watch on the left wrist and touches the Crown with a left-hand finger — or rests both hands on the same arm — there is no Lead I vector, the differential signal collapses, and the result will be inconclusive or noisy. The watch is not “broken” when this happens; the geometry simply fails to form.
The signal chain from skin to label
What the watch does between contact and result is the part every consumer guide skips. The voltage difference at the electrodes is on the order of 1 mV — three orders of magnitude smaller than the mains-frequency hum a human body picks up just standing in a room. Apple’s hardware design follows a pipeline any biosignal engineer would recognize, with the constraint that everything must run on a battery-powered watch.
The two electrodes feed an instrumentation amplifier that takes their difference and rejects the common-mode signal both electrodes see equally — the body acting as an antenna for 50 or 60 Hz mains hum. A high-resolution analog-to-digital converter then samples the differential signal; the export format published in Apple’s HKElectrocardiogram developer documentation shows samples consistent with a 512 Hz rate, well above the Nyquist threshold for the ~150 Hz upper edge of clinical ECG content. A digital filter chain follows: a high-pass around 0.5 Hz to strip baseline wander from breathing, a low-pass around 40 Hz to suppress muscle EMG, and a notch around 50/60 Hz for residual mains noise. Only after that conditioning does the rhythm classifier see the signal.
Related: a watchOS 26.2 sensor bug.
The classifier itself is the part Apple discloses least and the part that does the medical-device work. According to the DEN180044 decision summary, it operates on R-R interval irregularity and P-wave morphology, returning a confidence-gated label. Confidence below threshold returns “inconclusive” — by design, the algorithm refuses rather than guesses. Heart rates outside a defined window are returned as a rate result instead of a rhythm label, because the AFib classifier was validated only inside that window.
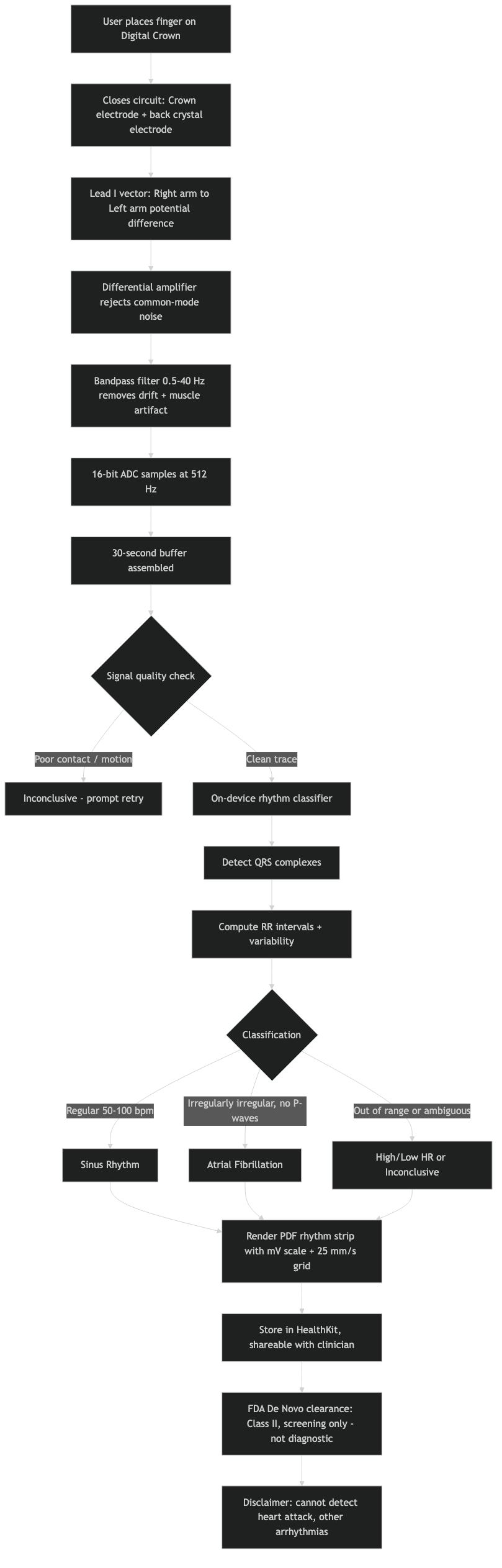
The architecture diagram above traces the full signal flow: skin → instrumentation amplifier → ADC → DSP filter chain → AFib classifier → label. Each stage exists for a specific reason. The amplifier provides common-mode rejection, the ADC sets dynamic range and quantization, the filters define what counts as signal, and the classifier handles only the binary AFib-vs-sinus decision Apple cleared. Anything outside that decision space is not a missing feature; it is out-of-label use.
What DEN180044 actually cleared — and what it didn’t
Reading the FDA decision summary directly is the only way to know what the device is legally allowed to claim. The De Novo DEN180044 decision, granted September 11, 2018, classifies the ECG App as a Class II software-only mobile medical app intended to “create, record, store, transfer, and display a single channel electrocardiogram (ECG) similar to a Lead I ECG” and to “determine the presence of atrial fibrillation (AFib) or sinus rhythm on a classifiable waveform.”
Three pieces of fine print do most of the interpretive work:
There is a longer treatment in life-saving health features.
- Indications for use: over-the-counter, ages 22 and older, classifiable waveforms only.
- Contraindications: not recommended for users with known arrhythmias other than AFib; not for use with pacemakers or implantable cardioverter defibrillators.
- Disclaimers: the data is informational; users should not interpret or act on it without a qualified healthcare professional; the waveform supplements, not replaces, traditional diagnostic methods.
A second clearance — K201525 — extended the upper detection limit from 120 BPM to 150 BPM, which is why the v2 algorithm tolerates faster rates without bouncing to a “high heart rate” non-result. Everything outside this label is, by Apple’s own filing, out of scope: heart attack detection, stroke risk, blood clots, premature ventricular contractions, atrial flutter, second- and third-degree AV blocks, and ventricular tachycardia all sit outside the cleared indication. When people complain that the watch “missed” their flutter or PVCs, they are describing the cleared scope, not a defect.
PPG vs ECG: two sensors, two features, one common confusion
Most consumer-facing coverage treats “Apple Watch heart monitoring” as one thing. It is two distinct features, on two different sensors, cleared under two different FDA submissions, with very different limitations.
The optical heart sensor on the back of the watch uses photoplethysmography (PPG) — green and infrared LEDs flash through the skin while photodiodes measure reflected light, varying as blood volume in the wrist pulses with each heartbeat. PPG runs passively and continuously. When its rhythm-analysis algorithm sees enough irregularly-spaced pulses across multiple background readings, it issues an “irregular rhythm notification.” That feature was cleared separately under De Novo DEN180042, also in September 2018.
A related write-up: wrist-based health monitoring.
The ECG app uses the electrical sensor and only runs when you start a recording and hold a finger on the Crown. It produces an actual waveform — a voltage-over-time tracing showing P-QRS-T complexes. PPG produces a pulse, not a waveform; it tells you when blood arrived at the wrist, not what the atria and ventricles were doing electrically.
The practical consequence: an irregular rhythm notification is not an AFib diagnosis, and the watch does not deliver one without your finger. That is why Apple’s ECG instructions direct users who receive an optical irregular-rhythm alert to open the ECG app and record a strip. Two features, in series — passive screen, then on-demand confirmation.
Why “inconclusive” is the correct answer
The most-debated number in the consumer literature is the unclassified rate. Pepplinkhuizen and colleagues, in a 2022 study published in Cardiovascular Digital Health Journal, ran 129 Apple Watch ECG recordings on patients undergoing electrical cardioversion and reported that 27.9% of first attempts came back unclassified. Once a recording was classifiable, sensitivity was 93.5% and specificity 100%. The headline reads as a failure rate; it is better read as a calibration choice.
Several conditions force “inconclusive”: a heart rate outside the cleared window, signal noise above a confidence threshold, paced beats, and morphologies the classifier was not trained to recognize. Atrial flutter at 2:1 conduction often looks like sinus rhythm to a single-lead algorithm because the second flutter wave hides inside the QRS. The same study found that allowing one repeat brought unclassified results down meaningfully without harming accuracy, while a third attempt began producing false positives. That gives a usable rule:
For more on this, see broader public-health context.
- If the first recording is inconclusive, repeat once with both feet flat, both arms supported, and the watch hand resting on a non-conductive surface.
- If the second is also inconclusive, stop. Repeating further trades information for noise — and false positives appear by the third try.
- If a recording reports AFib, the classifier’s specificity is high but its scope is narrow. Confirm with a clinician who can order a 12-lead or 24-hour Holter before any treatment decision.
- Inconclusive plus symptoms (chest pain, syncope, severe shortness of breath) is a clinical situation, not a watch problem; the watch cannot rule out anything outside its label.
The Pepplinkhuizen number sits next to a much larger one. The Apple Heart Study with Stanford enrolled more than 419,000 participants. Only 0.5% received an irregular-rhythm notification, and among those who wore an ECG patch and received a notification simultaneously, 84% were in AFib at the time of alert. Those measurements describe different things: Pepplinkhuizen measures the ECG app on a known-AF cohort; the Apple Heart Study measures the optical algorithm in a general population. Both can be true at once.
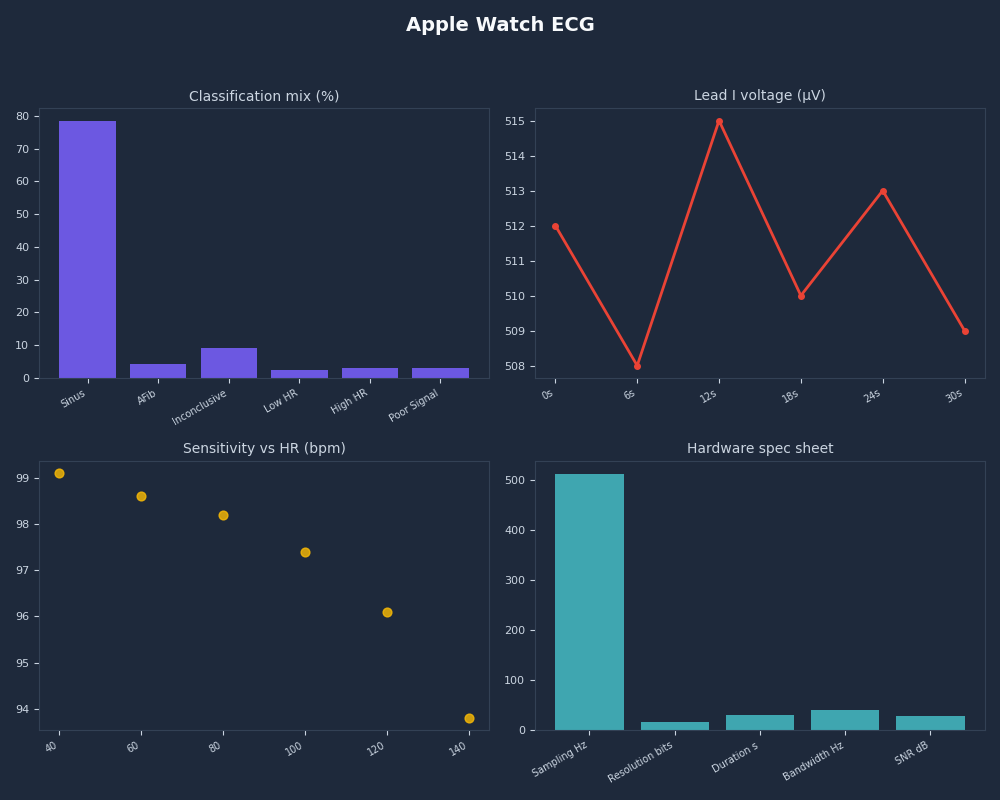
Strengths and gaps across Apple Watch ECG.
The dashboard above stitches these signals together. The lower the population prevalence of AFib, the lower any positive predictive value will go — that is base-rate arithmetic, not a sensor flaw. PPV in a 25-year-old marathoner is much lower than PPV in a screened 65-year-old cohort, even with an identical algorithm.
Decision framework: when to trust the watch, when to repeat, when to escalate
Reading the FDA label and the validation literature together gives a usable rubric for what the watch is — and isn’t — the right tool for. Treat it as a triage instrument, not a diagnostic.
- Pick the ECG app if you are 22 or older, have no pacemaker or ICD, and want an on-demand strip to bring to a clinician — for example, after a palpitation, an irregular-rhythm alert, or a self-monitoring request from your doctor.
- Pick the optical irregular-rhythm notification if you want passive, continuous background screening across days or weeks without remembering to record. Treat any alert as a prompt to open the ECG app, not as a diagnosis.
- Choose AFib History instead if you already have a clinician-confirmed AFib diagnosis and want to track time-in-AFib over a week. It does not screen new users — FDA labeling requires self-attested prior diagnosis to enable it.
- Repeat once when the first recording is inconclusive: sit still, feet flat, both arms supported, watch hand resting on a non-conductive surface. Pepplinkhuizen shows a single repeat recovers most classifiable signal without hurting accuracy.
- Stop repeating when the second attempt is also inconclusive. A third attempt begins producing false positives in the same study — you have hit the algorithm’s confidence floor and more samples will mislead, not clarify.
- Escalate to a 12-lead or 24-hour Holter when you have symptoms (chest pain, syncope, severe shortness of breath), a watch result you intend to act on, or any concern about a rhythm other than AFib. Flutter, PVCs, AV blocks, and ventricular tachycardia all sit outside the cleared label and the watch will not call them.
- Skip the ECG app entirely when you have a pacemaker or ICD (contraindicated), are under 22, or are looking for ischemia, MI, or any structural-heart finding. Lead I cannot localize ischemia and the classifier was not trained to flag it.
The shorthand: watch for screening, doctor for diagnosis, 12-lead for anything beyond AFib. The watch’s real value is capturing a tracing in the moment a symptom occurs — which a routine clinic ECG taken hours later usually misses.
Related: on-device health intelligence.
Reading a real strip: what the PDF export shows
Open the Health app on iPhone, tap an ECG entry, and “Export a PDF for Your Doctor” produces a one-page rhythm strip. The page carries information your watch screen does not. The 1 mV calibration pulse on the left edge sets the vertical scale; the time grid sets the horizontal scale at 25 mm/s, the international ECG standard. The classification line — sinus rhythm, AFib, low or high heart rate, inconclusive, or poor recording — sits above the trace, with the average heart rate and a metadata block. The sample rate is recorded in the export metadata and aligns with the 512 Hz figure in the developer documentation.
What a clinician reads off this strip that the watch label does not state: P-wave amplitude and morphology (whether atria are firing organized waves), PR interval (AV-node conduction), QRS duration (whether ventricular conduction is slowed or blocked), QT interval (a window onto repolarization risk that no smartwatch interprets), and any visible ectopy such as PACs or PVCs. The watch does not call any of this. It calls AFib or sinus, and a small number of error states. Everything else is the clinician’s job.
What changed Series 4 → Ultra 2, and what didn’t
The hardware that produces the Lead I tracing has not meaningfully changed since the Series 4 launched in 2018. The two electrodes, the differential amplifier, and the single-channel design are identical across every ECG-capable watch. What has changed sits in software.
| Model | Year | ECG hardware | Notable change relevant to ECG |
|---|---|---|---|
| Series 4 | 2018 | Lead I, two electrodes | ECG app v1, 50–120 BPM (DEN180044) |
| Series 5 | 2019 | Identical | Always-on display; ECG path unchanged |
| Series 6 | 2020 | Identical | Adds blood oxygen sensor; ECG path unchanged |
| Series 7 | 2021 | Identical | v2 algorithm extends rhythm window to 50–150 BPM (K201525) |
| Series 8 | 2022 | Identical | watchOS 9 introduces AFib History for diagnosed users |
| SE (1st/2nd gen) | 2020 / 2022 | No ECG sensor | Optical irregular-rhythm only |
| Ultra | 2022 | Identical Lead I | AFib History supported |
| Series 9 | 2023 | Identical | Double-tap gesture; ECG path unchanged |
| Ultra 2 | 2023 | Identical | Same Lead I sensor; AFib History |
| Series 10 | 2024 | Identical | Sleep apnea notifications |
Source: Apple support documentation and FDA submissions DEN180044 and K201525.
More detail in smartwatch health roadmap.
The line worth drawing under the table: AFib History, introduced in watchOS 9 in 2022, is not a new diagnostic. It is a longitudinal aggregation of optical readings for users who have already been diagnosed with AFib, presenting an estimate of the percentage of time spent in AFib over a week. It does not screen new users; FDA labeling is explicit that users must self-attest a prior diagnosis to enable it. The hardware is a 2018 part doing 2018 work; the software is what has accumulated.
If you want a single working model of the device, treat the Apple Watch ECG as a screening AFib detector with one input geometry, two failure-tolerant repeat attempts, and an explicit refusal mode for ambiguous cases. It is not a 12-lead. It will not detect what it was not trained to detect. The “limitations” people complain about are the boundaries of the cleared label — and the cleared label is what makes it a medical device instead of a wellness toy. Read the label, repeat once if inconclusive, and bring the PDF to a clinician for anything that matters.
Continue with untethered cellular Watch future.
Further reading
- FDA De Novo decision summary DEN180044 — ECG App
- FDA De Novo decision summary DEN180042 — Irregular Rhythm Notification Feature
- FDA 510(k) clearance K201525 — extended rhythm window to 150 BPM
- Pepplinkhuizen et al., “Accuracy and clinical relevance of the single-lead Apple Watch electrocardiogram to identify atrial fibrillation,” Cardiovascular Digital Health Journal, 2022
- Stanford Medicine: Apple Heart Study results (419,000+ participants)
- Apple Developer Documentation — HKElectrocardiogram export format
- Apple Support — Take an ECG with the ECG app on Apple Watch